Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tsuji, Hayato*; Nakahata, Masaki*; Hishida, Mafumi*; Seto, Hideki*; Motokawa, Ryuhei; Inoue, Takeru*; Egawa, Yasunobu*
Journal of Physical Chemistry Letters (Internet), 14(49), p.11235 - 11241, 2023/12
Times Cited Count:0 Percentile:0.01(Chemistry, Physical) neutron diffraction
neutron diffractionKobayashi, Hiroki*; Komatsu, Kazuki*; Ito, Hayate*; Machida, Shinichi*; Hattori, Takanori; Kagi, Hiroyuki*
Journal of Physical Chemistry Letters (Internet), 14(47), p.10664 - 10669, 2023/11
Times Cited Count:1 Percentile:0.01(Chemistry, Physical)Ice IV is a metastable high-pressure phase of ice in which the water molecules exhibit orientational disorder. Although orientational ordering is commonly observed for other ice phases, it has not been reported for ice IV. We conducted  powder neutron diffraction experiments for DCl-doped D
powder neutron diffraction experiments for DCl-doped D O ice IV to investigate hydrogen ordering in ice IV. We found abrupt changes in the temperature derivative of unit cell volume, dV/dT, at about 120 K, and revealed their slightly ordered structure at low temperatures based on the Rietveld method. The occupancy of the D1 site deviates from 0.5; it increased when samples were cooled at higher pressures and reached 0.282(5) at 2.38 GPa, 58 K. Our results evidence the presence of a low-symmetry hydrogen-ordered state corresponding to ice IV. It seems, however, difficult to experimentally access the completely ordered phase corresponding to ice IV by slow cooling at high pressure.
O ice IV to investigate hydrogen ordering in ice IV. We found abrupt changes in the temperature derivative of unit cell volume, dV/dT, at about 120 K, and revealed their slightly ordered structure at low temperatures based on the Rietveld method. The occupancy of the D1 site deviates from 0.5; it increased when samples were cooled at higher pressures and reached 0.282(5) at 2.38 GPa, 58 K. Our results evidence the presence of a low-symmetry hydrogen-ordered state corresponding to ice IV. It seems, however, difficult to experimentally access the completely ordered phase corresponding to ice IV by slow cooling at high pressure.
Kumada, Takayuki; Nakagawa, Hiroshi; Miura, Daisuke; Sekine, Yurina; Motokawa, Ryuhei; Hiroi, Kosuke; Inamura, Yasuhiro; Oku, Takayuki; Oishi, Kazuki*; Morikawa, Toshiaki*; et al.
Journal of Physical Chemistry Letters (Internet), 14(34), p.7638 - 7643, 2023/08
Times Cited Count:0 Percentile:0.01(Chemistry, Physical)The structure of nano-ice crystals in rapidly frozen glucose solution was elucidated by using spin-contrast-variation small-angle neutron scattering, which distinguishes the nano-ice crystal signal from the frozen amorphous solution signal by the polarization-dependent neutron scattering. The analysis revealed that the nano-ice crystals form a planar structure with a diameter exceeding tens of nanometers and a thickness of 1 nm, which is close to the critical nucleation size. This result suggests that the glucose molecules are preferentially bound to a specific face of nano-ice crystals, and then block the crystal growth perpendicular to that face.
 ortho-para conversion on a metal surface; Interplay between electron and phonon systems
ortho-para conversion on a metal surface; Interplay between electron and phonon systemsUeta, Hirokazu; Fukutani, Katsuyuki
Journal of Physical Chemistry Letters (Internet), 14(34), p.7591 - 7596, 2023/08
Times Cited Count:1 NH and CaNH thin films by reactive magnetron sputtering under hydrogen partial pressure control
NH and CaNH thin films by reactive magnetron sputtering under hydrogen partial pressure controlChon, S.*; Fukutani, Katsuyuki; 8 of others*
Journal of Physical Chemistry Letters (Internet), 13(43), p.10169 - 10174, 2022/11
Times Cited Count:0 Percentile:0.01(Chemistry, Physical)Kusaka, Ryoji; Watanabe, Masayuki
Journal of Physical Chemistry Letters (Internet), 13(30), p.7065 - 7071, 2022/08
Times Cited Count:5 Percentile:67.52(Chemistry, Physical) Reactions to Achieve Mass-spectrometric Discrimination in Simultaneous Plutonium-isotope Speciation with Inductively Coupled Plasma-Tandem Mass Spectrometry
Reactions to Achieve Mass-spectrometric Discrimination in Simultaneous Plutonium-isotope Speciation with Inductively Coupled Plasma-Tandem Mass SpectrometryMatsueda, Makoto; Kawakami, Tomohiko*; Koarai, Kazuma; Terashima, Motoki; Fujiwara, Kenso; Iijima, Kazuki; Furukawa, Makoto*; Takagai, Yoshitaka*
Chemistry Letters, 51(7), p.678 - 682, 2022/07
Times Cited Count:6 Percentile:58.73(Chemistry, Multidisciplinary)New methodology for a simultaneous isotope speciation of various Pu isotopes without complicated isobaric interferences is developed by using inductively coupled plasma-mass spectrometry (ICP-MS). In analyzing ICP tandem MS (ICP-MS/MS), CO gas reactions in a dynamic reaction cell (DRC) almost eliminated the background noise intensity produced by isobaric interference from isotopes originating from actinides such as Am, Cm, and U at the locations (m/z) of significant Pu isotopes (
gas reactions in a dynamic reaction cell (DRC) almost eliminated the background noise intensity produced by isobaric interference from isotopes originating from actinides such as Am, Cm, and U at the locations (m/z) of significant Pu isotopes ( Pu,
Pu, 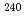 Pu,
Pu,  Pu,
Pu, 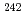 Pu,
Pu,  Pu).
Pu).
 microphase separation
microphase separationIsozaki, Yuka*; Higashiharaguchi, Seiya*; Kaneko, Naoya*; Yamazaki, Shun*; Taniguchi, Tatsuo*; Karatsu, Takashi*; Ueda, Yuki; Motokawa, Ryuhei
Chemistry Letters, 51(6), p.625 - 628, 2022/06
Times Cited Count:2 Percentile:27.64(Chemistry, Multidisciplinary)Wang, X.*; Tang, X.*; Zhang, P.*; Wang, Y.*; Gao, D.*; Liu, J.*; Hui, K.*; Wang, Y.*; Dong, X.*; Hattori, Takanori; et al.
Journal of Physical Chemistry Letters (Internet), 12(50), p.12055 - 12061, 2021/12
Times Cited Count:6 Percentile:50.99(Chemistry, Physical)Substituted polyacetylene is expected to improve the chemical stability, physical properties, and additional functions of the polyacetylene backbones, but its diversity is very limited. Here, by applying external pressure on solid acetylenedicarboxylic acid, we report the first crystalline poly-dicarboxylacetylene with every carbon on the trans-polyacetylene backbone bonded to a carboxyl group, which is very hard to synthesize by traditional methods. This unique structure combines the extremely high content of carbonyl groups and high conductivity of a polyacetylene backbone, which exhibits a high specific capacity and excellent cycling/rate performance as a Li-ion battery (LIB) anode. We present a completely functionalized crystalline polyacetylene and provide a high-pressure solution for the synthesis of polymeric LIB materials and other polymeric materials with a high content of active groups.
Kaneko, Masashi; Sasaki, Yuji; Wada, Eriko*; Nakase, Masahiko*; Takeshita, Kenji*
Chemistry Letters, 50(10), p.1765 - 1769, 2021/10
Times Cited Count:0 Percentile:0(Chemistry, Multidisciplinary)Density functional theory calculation is applied to predict the stability constants for Eu and Am
and Am complexes in aqueous solution for molecular modelling of novel separation agents for minor actinides over lanthanides. Logarithm of experimental stability constants correlates with calculated complex formation enthalpies with high reproducibility (R
complexes in aqueous solution for molecular modelling of novel separation agents for minor actinides over lanthanides. Logarithm of experimental stability constants correlates with calculated complex formation enthalpies with high reproducibility (R
 0.98). Prediction of stability constants of novel chelates is demonstrated and indicates a potential availability of the derivatives of diethylenetriaminepentaacetic acid type chelate in acidic condition and enhancement of Am
0.98). Prediction of stability constants of novel chelates is demonstrated and indicates a potential availability of the derivatives of diethylenetriaminepentaacetic acid type chelate in acidic condition and enhancement of Am selectivity over Eu
selectivity over Eu .
.
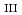 -edge for the determination of oxidation state for trace amount of Eu in natural samples by bragg-type crystal analyzer system
-edge for the determination of oxidation state for trace amount of Eu in natural samples by bragg-type crystal analyzer systemKonagaya, Rimi*; Kawamura, Naomi*; Yamaguchi, Akiko; Takahashi, Yoshio*
Chemistry Letters, 50(8), p.1570 - 1572, 2021/08
Times Cited Count:3 Percentile:22.03(Chemistry, Multidisciplinary)no abstracts in English
Ouchi, Kazuki; Komatsu, Atsushi; Takao, Koichiro*; Kitatsuji, Yoshihiro; Watanabe, Masayuki
Chemistry Letters, 50(6), p.1169 - 1172, 2021/06
Times Cited Count:2 Percentile:14.19(Chemistry, Multidisciplinary)The electrochemical behavior of uranium (IV) tetrachloride in ionic liquid-DMF mixture was studied for first time in order to build a redox flow battery (RFB) using U as an electrode active material. We found a quasi-reversible U /U
/U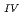 couple that could be applied to the anode reaction of the RFB.
couple that could be applied to the anode reaction of the RFB.
Yamamoto, Naoki*; Kofu, Maiko; Nakajima, Kenji; Nakagawa, Hiroshi; Shibayama, Naoya*
Journal of Physical Chemistry Letters (Internet), 12(8), p.2172 - 2176, 2021/03
Times Cited Count:9 Percentile:67.44(Chemistry, Physical)Hydration water plays a crucial role for activating the protein dynamics required for functional expression. Yet, the details are not understood about how hydration water couples with protein dynamics. A temperature hysteresis of the ice formation of hydration water is a key phenomenon to understand which type of hydration water, unfreezable or freezable hydration water, is crucial for the activation of protein dynamics. Using neutron scattering, we observed a temperature-hysteresis phenomenon in the diffraction peaks of the ice of freezable hydration water, whereas protein dynamics did not show any temperature hysteresis. These results show that the protein dynamics is not coupled with freezable hydration water dynamics, and unfreezable hydration water is essential for the activation of protein dynamics.
 at the structure factor primary peak and prepeak
at the structure factor primary peak and prepeakLuo, P.*; Zhai, Y.*; Leao, J. B.*; Kofu, Maiko; Nakajima, Kenji; Faraone, A.*; Zhang, Y.*
Journal of Physical Chemistry Letters (Internet), 12(1), p.392 - 398, 2021/01
Times Cited Count:4 Percentile:35.04(Chemistry, Physical)Using neutron spin-echo spectroscopy, we studied the microscopic structural relaxation of a prototypical network ionic liquid ZnCl at the structure factor primary peak and prepeak. The results show that the relaxation at the primary peak is faster than the prepeak and that the activation energy is
at the structure factor primary peak and prepeak. The results show that the relaxation at the primary peak is faster than the prepeak and that the activation energy is 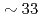 % higher. A stretched exponential relaxation is observed even at temperatures well-above the melting point
% higher. A stretched exponential relaxation is observed even at temperatures well-above the melting point 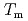 . Surprisingly, the stretching exponent shows a rapid increase upon cooling, especially at the primary peak, where it changes from a stretched exponential to a simple exponential on approaching the
. Surprisingly, the stretching exponent shows a rapid increase upon cooling, especially at the primary peak, where it changes from a stretched exponential to a simple exponential on approaching the 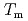 . These results suggest that the appearance of glassy dynamics typical of the supercooled state even in the equilibrium liquid state of ZnCl
. These results suggest that the appearance of glassy dynamics typical of the supercooled state even in the equilibrium liquid state of ZnCl as well as the difference of activation energy at the two investigated length scales are related to the formation of a network structure on cooling.
as well as the difference of activation energy at the two investigated length scales are related to the formation of a network structure on cooling.
Ogawa, Shuichi*; Yamaguchi, Hisato*; Holby, E. F.*; Yamada, Takatoshi*; Yoshigoe, Akitaka; Takakuwa, Yuji*
Journal of Physical Chemistry Letters (Internet), 11(21), p.9159 - 9164, 2020/11
Times Cited Count:3 Percentile:17.73(Chemistry, Physical)Atomically thin layers of graphene have been proposed to protect surfaces through the direct blocking of corrosion reactants such as oxygen with low added weight. The long term efficacy of such an approach, however, is unclear due to the long-term desired protection of decades and the presence of defects in as-synthesized materials. Here, we demonstrate catalytic permeation of oxygen molecules through previously-described impermeable graphene by imparting sub-eV kinetic energy to molecules. These molecules represent a small fraction of a thermal distribution thus this exposure serves as an accelerated stress test for understanding decades-long exposures. The permeation rate of the energized molecules increased 2 orders of magnitude compared to their non-energized counterpart. Graphene maintained its relative impermeability to non-energized oxygen molecules even after the permeation of energized molecules indicating that the process is non-destructive and a fundamental property of the exposed material.
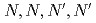 -tetraoctyl-diglycolamide from organic acid
-tetraoctyl-diglycolamide from organic acidSasaki, Yuji; Matsumiya, Masahiko*; Nakase, Masahiko*; Takeshita, Kenji*
Chemistry Letters, 49(10), p.1216 - 1219, 2020/10
Times Cited Count:9 Percentile:44.36(Chemistry, Multidisciplinary)Lanthanide (Ln) extractions from organic acids to  -dodecane by
-dodecane by 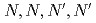 -tetraoctyl-diglycolamide (TODGA) were conducted. Four organic acids (lactic acid, malonic acid, tartaric acid, and citric acid) were employed. Although these acids stabilize lanthanides in the aqueous phase, a distribution ratio (
-tetraoctyl-diglycolamide (TODGA) were conducted. Four organic acids (lactic acid, malonic acid, tartaric acid, and citric acid) were employed. Although these acids stabilize lanthanides in the aqueous phase, a distribution ratio ( ) greater 1 was obtained for heavy Ln. Ln patterns (
) greater 1 was obtained for heavy Ln. Ln patterns ( (Ln) against atomic number of Ln) show maximum values of Ho and Er. In order to obtain high
(Ln) against atomic number of Ln) show maximum values of Ho and Er. In order to obtain high  values, the addition of HNO
values, the addition of HNO in aqueous phase is found to be effective.
in aqueous phase is found to be effective.
Gonzal z, M. A.*; Borodin, O.*; Kofu, Maiko; Shibata, Kaoru; Yamada, Takeshi*; Yamamuro, Osamu*; Xu, K.*; Price, D. L.*; Saboungi, M.-L.*
z, M. A.*; Borodin, O.*; Kofu, Maiko; Shibata, Kaoru; Yamada, Takeshi*; Yamamuro, Osamu*; Xu, K.*; Price, D. L.*; Saboungi, M.-L.*
Journal of Physical Chemistry Letters (Internet), 11(17), p.7279 - 7284, 2020/09
Times Cited Count:17 Percentile:78.45(Chemistry, Physical)Nagasawa, Makoto*; Qin, H.-B.*; Yamaguchi, Akiko; Takahashi, Yoshio*
Chemistry Letters, 49(8), p.909 - 911, 2020/08
Times Cited Count:3 Percentile:15.14(Chemistry, Multidisciplinary) -olefin using small-angle X-ray scattering
-olefin using small-angle X-ray scatteringOba, Yojiro; Motokawa, Ryuhei; Hino, Masahiro*; Adachi, Nozomu*; Todaka, Yoshikazu*; Inoue, Rintaro*; Sugiyama, Masaaki*
Chemistry Letters, 49(7), p.823 - 825, 2020/07
Times Cited Count:0 Percentile:0(Chemistry, Multidisciplinary) /Y
/Y Fe
Fe O
O layers via longitudinal spin Seebeck effect
layers via longitudinal spin Seebeck effectLee, W.-Y.*; Park, N.-W.*; Kang, M.-S.*; Kim, G.-S.*; Jang, H. W.*; Saito, Eiji; Lee, S.-K.*
Journal of Physical Chemistry Letters (Internet), 11(13), p.5338 - 5344, 2020/07
Times Cited Count:11 Percentile:62.94(Chemistry, Physical)